|
Once you collect your sample, you simply mail the sample in according to the directions that are in the kit. Patient guide instructions and how to return your kit pamphlet.Opening & Using The KitĮach Cologuard kit contains the following: The lab will return the results to your physician or healthcare provider who will contact you as soon as they arrive. Once you collect a sample of your stool, you will send or drop off your sample to a lab. You are not required to change your diet or medications before this test. The Cologuard home colon cancer screening test detects cancer by identifying DNA markers and occult blood in stool samples. The test is highly sensitive to microscopic amounts of blood caused by these bleeding polyps. A polyp is an abnormal growth of tissue that often will bleed due to tiny broken blood vessels caused by the stool passing through the colon. The test specifically tests for hemoglobin, the iron-containing protein that carries the oxygen in the blood cell. Stool samples are also tested for the presence of a protein found in blood using the antibody test, fecal immunochemical test (FIT). These biomarkers indicate the presence of colorectal cancer or precancerous tumors known as adenomas. The test detects mutations in the following genes: Lab pathologists test the stool samples sent in by patients for increased levels of mutated DNA biomarkers that are found in shedded colon cells. The Cologuard home colon cancer screening test detects cancer by identifying DNA markers from abnormal cells and occult blood in stool samples. A cancerous tumor in the colon will contain mutated DNA fragments that can be detected with special lab tests like the FIT- DNA test or Cologuard. Abnormal cells from a polyp or tumor will also shed into the feces along with any occult blood caused by broken blood vessels. This shedding allows the intestinal lining to renew and maintain its healthy tissue for optimal health. The lining of the colon sheds cells routinely throughout the day and these cells end up in the feces, or stool. The Cologuard screening test is a combination of two tests, the fecal immunochemical test (FIT) and a DNA screening for abnormal cancer cells. If there are any polyps found or abnormal tissue growths (adenomas), you may not be eligible for home screenings from that point on.Ĭologuard: A Combination of Two Tests FIT-DNA Keep in mind that if you test positive in any of the home screening tests, you will need a colonoscopy to locate the source of the bleed and get any associated polyps removed. Because of these accurate ratings, Cologuard tests can be repeated every three years in average risk patients. Cologuard: Has a specificity of 95% for ruling out the presence of polyps and colon cancerĬologuard is highly sensitive due to its ability to identify cancerous cells using DNA-detecting technology.Cologuard: 94% of early stage colon cancers are sensitive to the Cologuard test.Cologuard: Has a 92.3% sensitivity for detecting colon cancer at all stages.Important performance findings of Cologuard tests include: very small polyps and the precancerous adenomas. A positive result from a Cologuard screening is generally followed by a colonoscopy to rule out colorectal cancer and to remove any polyps that may be found during the visual exploration of the colon. Cologuard also detects microscopic amounts of blood in the stool, so it serves as a dual screening for both mutated cells shed by cancerous tumors and bleeding polyps. The test detects mutations in the colon cells that shed from the intestinal lining into the feces. 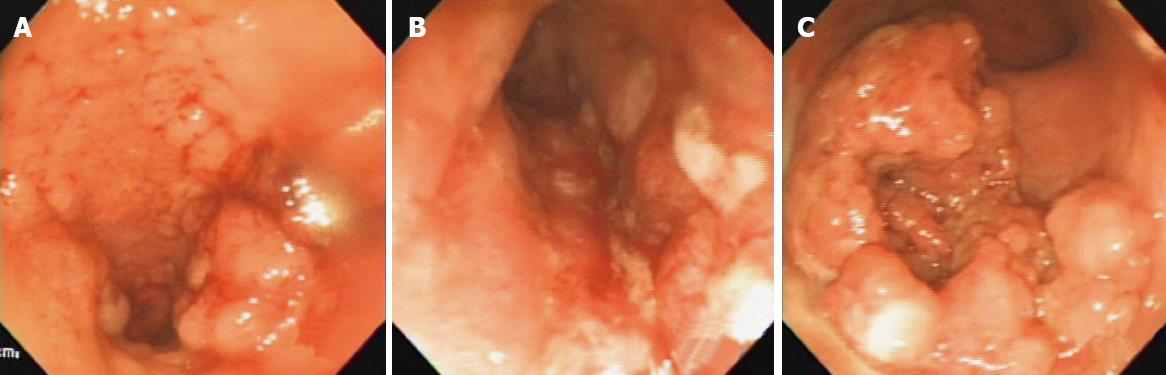
OverviewĬologuard, also known as a multitarget DNA stool test, is a non-invasive colon cancer screening test that is done in the privacy of your home and mailed to a lab for analysis You are eligible for Cologuard if you are at an average risk for colorectal cancer with no family or personal history of colon cancer. In 2019, Exact Sciences labs processed 1.69 million Cologuard tests while touting its 92% sensitivity and 87% specificity rates for detecting colorectal cancer as selling points.Ĭologuard, also known as a multitarget DNA stool test, is a non-invasive colon cancer screening test that is done in the privacy of your home and mailed to a lab for analysis You are eligible for Cologuard if you are at an average risk for colorectal cancer with no family or personal history of colon cancer. Since its release into the medical community, approximately 3.5 million people have used Cologuard to screen for colorectal cancer. The company responsible for the invention of Cologuard is Exact Sciences, a multimillion dollar research and development conglomerate that specializes in at-home cancer screening tests. In August of 2014, the FDA approved Cologuard the first colorectal screening test that uses DNA to identify cancer cells in a patient’s stool sample.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
